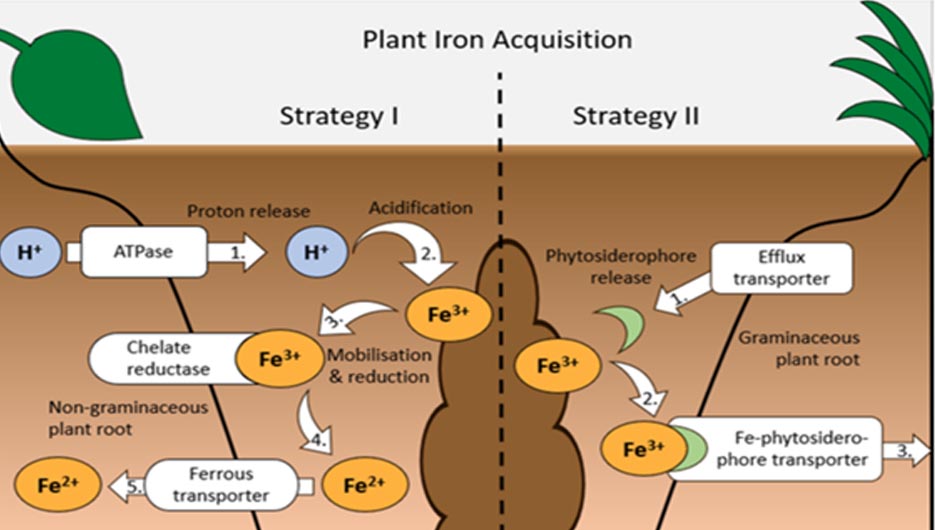
Two factors can interfere with acquisition and use of Fe by crops, even when Fe availability in soils is not limiting. Firstly, plant breeding to increase crop yield and improve resistance to pathogens or pests can select for traits that increase the plant’s micronutrient requirements, creating Fe deficiencies, even though soil Fe availability was considered sufficient for past cultivars. Secondly, the application of herbicides such as glyphosate, diclofop-methyl and chlorsulfuron can interfere with root growth and Fe translocation from the roots to shoots and grain, resulting in plant Fe deficiency. Plants employ two strategies to improve Fe acquisition under deficient conditions. Strategy I, used by nongraminaceous monocots and dicots (e.g., legumes), involves the release of protons into the rhizosphere, which increases soil acidity and mobilizes ferric Fe(III). Enzymes (chelate reductases) at the root–soil interface then reduce Fe(III) to ferrous Fe(II), which can be absorbed by the plant via ferrous transporters. Strategy II plants are graminaceous monocots (e.g., grasses and cereal crops); the plants exude Fe-chelating organic substances (phytosiderophores), which form complexes with Fe(III) that plants can then absorb via plasma membrane transport systems without reduction.
Denton-Thompson, S. Sayer, E. Soil Systems 6(1), 8 (2022)
https://doi.org/10.3390/soilsystems6010008
Photo: Diagram of the two iron (Fe) acquisition strategies employed by plants: orange ellipses show the chemical form of Fe, blue circles represent hydrogen (H+) ions, white boxes with arrows indicate molecule transport points into/out of the root, and numbers indicate individual steps of the process.


